Sodium Alginate
| Home | | Pharmacognosy |Chapter: Pharmacognosy and Phytochemistry : Drugs Containing Carbohydrates and Derived Products
Sodium alginate is the sodium salt of alginic acid. Alginic acid is a polyuronic acid composed of reduced mannuronic and glucoronic acids, which are obtained from the algal growth of the species of family Phaeophyceae. The common species are Macrocystis pyrifera, Laminaria hyperborea, Lami-naria digitata, Ascophyllum nodosum and Durvillaea lessonia. It is a purified carbohydrate extracted from brown seaweed (algae) by treatment of dilute alkali.
SODIUM ALGINATE
Synonyms
Algin, Alginic acid sodium salt, Sodium polymannuronate,
Kelgin, Minus, Protanal.
Biological Source
Sodium alginate is the sodium salt of alginic acid. Alginic
acid is a polyuronic acid composed of reduced mannuronic and glucoronic acids,
which are obtained from the algal growth of the species of family Phaeophyceae.
The common species are Macrocystis
pyrifera, Laminaria hyperborea, Lami-naria digitata, Ascophyllum nodosum and Durvillaea lessonia. It is a purified carbohydrate extracted
from brown seaweed (algae) by treatment of dilute alkali.
Geographical Source
Sea-weeds are found in Atlantic and Pacific oceans,
par-ticularly in coastal lines of Japan, United States, Canada, Australia and
Scotland. In India, it is found near the coast of Saurashtra. The largest
production of algin is in United States and U.K.
Collection and Preparation
The brown coloured algae are used for extraction of alginic
acid. The colour is due to carotenoid pigment present in it. M. pyrifera, the principal source for
global supply, is a perennial plant that lives from 8 to 12 years, and grows,
as much as, 30 cm per day. This giant kelp is found mainly in Pacific Ocean. It
grows on stands from 15 m to 1.5 km in width and several km in length. The
mechanical harvesting is done about four times a year.
Alginic acid is present in the cell wall. The seaweeds are
harvested, dried, milled and extracted with dilute sodium carbonate solution
which results in a pasty mass. It is then diluted to separate insoluble matter.
Soft water is only used for extraction purposes, so as to avoid
incompatibilities. It is treated with calcium chloride or sulphuric acid for
conversion into either calcium alginate or insoluble alginic acid, which is
collected and purified by thorough washing. If calcium is used, it is treated
with hydrochloric acid. Alginic acid so collected is treated with sodium
carbonate for neutralization and converted into sodium salt. The alginic acid
content on dry solid basis varies from 22% to 35% in all the varieties of brown
algae.
Morphology
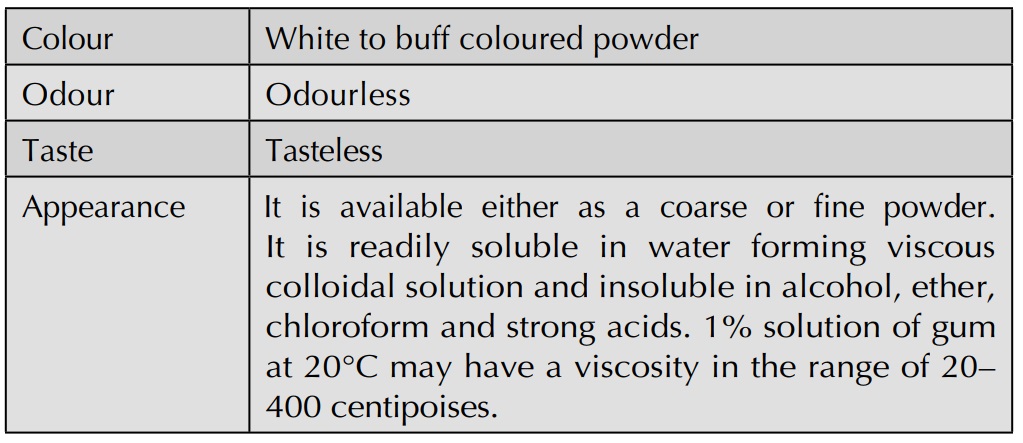
History
Alginic acid, a hard, horny polysaccharide, was first
isolated by the English chemist Stanford in 1883 and in Britain was first
marketed in 1910. The commercial production of algin first began in 1929 in
United States Since then it is produced in U.K., France, Norway and Japan. The
present total algin production is estimated to be more than 15,000 tones per
annum.
Identification Tests
1) Precipitate formation with Calcium chloride
To a 0.5%
solution of the sample in sodium hydroxide, add one-fifth of its volume of a
2.5% solution of calcium chloride. A voluminous, gelatinous precipitate is
formed. This test distinguishes sodium alginate from gum arabic, sodium
carboxymethyl cellulose, carrageenan, gelatin, gum ghatti, karaya gum and
tragacanth gum.
2) Precipitate formation with Ammonium
sulphate
To a 0.5%
solution of the sample in sodium hydroxide, add one-half of its volume of a
saturated solution of ammonium sulphate. No precipitate is formed. This test
distinguishes sodium alginate from agar, sodium carboxymethyl cellulose,
carrageenan, methyl cellulose and starch.
3) Test for alginate
Moisten 1–5
mg of the sample with water and add 1 ml of acid ferric sulphate. Within 5 min,
a cherry-red colour develops that finally becomes deep purple.
1% solution
in water forms heavy gelatinous precipitate with dilute sulphuric acid.
Chemical Constituents
Algin consists chiefly of the sodium salt of alginic acid, a
linear polymer of L-guluronic acid and D-mannuronic acid; the chain length is
long and varies (mol. wt. from 35,000 to 1.5 × 106) with the method of
isolation and the source of the algae. Mannuronic acid is the major component..
The alginic acid molecule appears to be a copolymer of 1, 4-linked
mannopyranosyluronic acid units, of 1, 4-linked gulopyranosyluronic acid units,
and of segments where these uronic acids alternate with 1, 4-linkages.
Uses
High and medium viscosity grades of sodium alginate are used
in the preparation of paste, creams and for thickening and stabilizing
emulsions. It is a good suspending and thickening agent, but a poor emulsifying
agent. It is used as binding and disintegrating agent in tablets and lozenges.
In food industry, it is used for the preparation of jellies, ice cream, etc. It
is also used in textile industry. For pharmaceutical purposes, when desired,
it is sterilized by heating in an autoclave. The solution of sodium alginate
should not be stored in metal containers. It is preserved by the addition of
0.1% of chloroxylenol, chlorocresol, benzoic acid or parabenes. Potassium,
aluminium and calcium alginates are also used medicinally.
Capsules containing sodium alginate and calcium carbon-ate
are used to protect inflamed areas near the entrance to the stomach. The
acidity of the stomach causes formation of insoluble alginic acid and carbon
dioxide; the alginic acid rises to the top of the stomach contents and forms a
protective layer.
Marketed Product
Each 100 ml of Lamina G solution manufactured by Taejoon
Pharm Co. Ltd, Seoul contains 5.0g of sodium alginate. It is mainly used for
the treatment of Gastric and duodenal ulcer, erosive gastritis, reflux
esophagitis (usual dosage is 20–60 ml orally three to four times daily before
meal) and Hemostasis in gastric biopsy (usual dosage is 10–30 ml by endoscope,
followed by 30 ml orally).
