Problems on Functional Groups and Chemical Bonding
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Functional Groups and Chemical Bonding
Organic Chemistry : Functional Groups and Chemical Bonding - Problems, Questions and answers on Functional Groups and Chemical Bonding
PROBLEMS
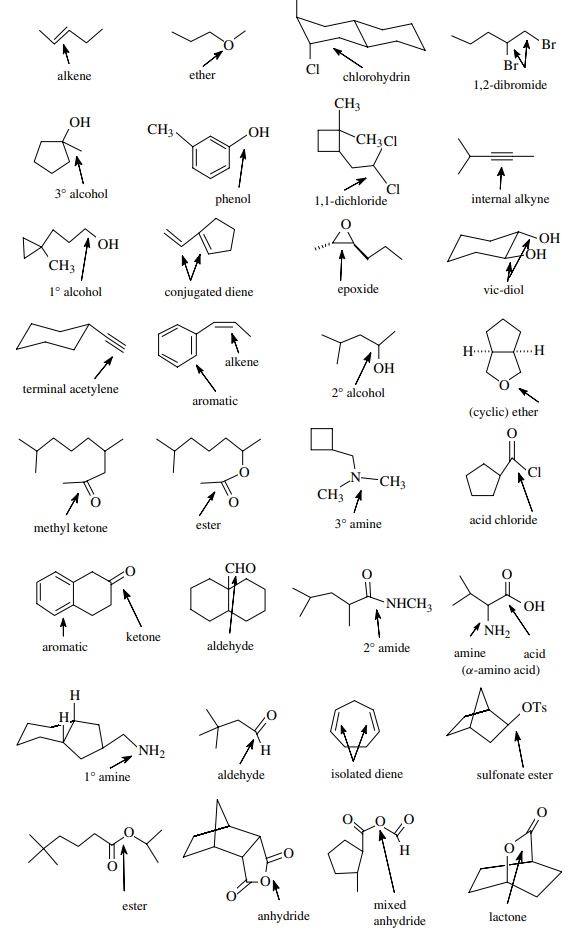
1 Excluding alkyl
groups, name and point out the functional groups in the following molecules:

Answer:
2. Give the bonding
scheme (orbitals, etc.) and geometry for the following functional groups:
(a) alkyl nitrile
(use R for alkyl group)
(b) alkyl azide (use
R for alkyl group)
(c) nitro alkane
(use R for alkyl group)
(d) N -methyl pyrrole (it is aromatic)
Answer:

3. For the following
compounds, give the approximate bond angles around the atoms indicated by an
arrow:

Answer:

4. For the following
compounds add all lone pairs of electrons to the struc-tures and then specify
the type of orbital in which they are located:

CH3OCH3
CH3CHO
CH3O−
CH3C ≡O+
CH3CH=NCH3
CH3CH2NH2
CH2=N−
CH3CN
CH3F
CH3Cl
CH3Br
CH3CNO
CH3NC
CH3SCN
CH3NCS
Answer:

5. On the basis of
electronic structure and orbital energies, supply predictions for the following
and explain your answer:
a. Which will be
more nucleophilic towards methyl iodide?

b. Which will be
more basic?

c. Which anion will
be more stable?

Answer:

Because
the electrons are in an sp2 orbital which has less s character than
the e− in the sp orbital
in the other compound.
(b)

Because
of being in an orbital of 25% s character compared to 33% s character in the
ketone, the e− are less tightly
bound and more easily donated to a proton.
(c)

Because the e− are in an sp orbital with greater s character than the lone pair in the sp2 orbital of the other compound.
6. Which of the
following compounds or ions are aromatic? Draw orbital diagrams to demonstrate
why.
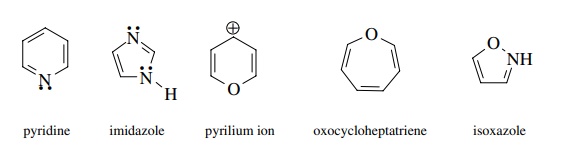
Answer:

7. Consider the
tropanyl anion T and the cyclopentadienyl anion C. Which one is more stable and
why? Predict the structure of each based on your analysis.

Answer:

8. Compound K is
found almost entirely in its enol form E. Why?

Answer:
Tautomer
K is a resonance-stabilized
structure as shown; however, tautomer E
has a principal resonance contributor which is an aromatic species. The need to
separate charges decreases the stability somewhat, but the aromatic system is a
large stabilizing feature. Thus E is
of much lower energy than K and the
equilibrium between K and E is shifted to E.

9. Explain why
compound B can be considered a doubly aromatic molecule.

Answer:

By
using the lone pair on nitrogen, each ring can have six π electrons and thus satisfy Huckel’s rule. Thus each ring is an
aromatic ring and the molecule can be considered doubly aromatic. The need to
separate charges to accomplish this decreases the aromatic stabilization
somewhat. Nevertheless the aromatic stabi-lization makes B’ a significant resonance contributor. As a consequence B has a great deal of charge separation
and thus a large molecular dipole moment of 9.6D.
10. It is found that
bromodiazirine A undergoes loss of bromide to produce a cation much more easily
than bromocyclopropane C. Can you think of a reason why?

Answer:

Both
bromides give benzylic cations which are resonance stabilized by the benzene
rings. The nitrogens of A are more
electronegative than the carbons of C
which should destabilize the ion somewhat. The greatest stabilization comes
from the fact that the three-membered cationic ring of A is a 2π aromatic
system. This aromatic stabilization of the cation makes its formation much more
rapid than the cation from C.
11. Compounds L1
and L2 are both lactones. Can you think of a reason why it is much
more difficult to remove the α proton
of L1 than to remove the α
proton of L2?

Answer:

Removal
of a proton from L1 gives
an anion whose lone pair is orthogonal to the π bond of the carbonyl group by virtue of the rigid geometry of the
bicyclic system. Consequently the lone pair cannot overlap with the carbonyl π bond and delocalization via resonance
is not possible — it is effectively a localized anion. Removal of a proton from
L2 gives rise to a lone
pair in a p orbital which can overlap with the carbonyl π bond and thus resonance delocalization is possible. Thus the
anion from L2 is
resonance stabilized and is thus formed more easily.
12. Explain why
squaric acid ionizes completely (dissociates two protons) in water and is
nearly as strong as sulfuric acid.

Answer:

The
bis anion of squaric acid is highly resonance stabilized by four equivalent
resonance contributors as well as an aromatic resonance contributor. This
results in both protons being easily removed.
13. Explain why
guanidine is one of the strongest noncharged organic bases known.

Answer:

Protonation
of guanidine gives an ion that is stabilized by four resonance forms, three of
which are equivalent. This gives a large amount of resonance stabilization to
the protonated form, making guanidine easily protonated and thus a good base.
14. Using resonance
arguments, explain why cyclopentadiene is more acidic than indene.

Answer:

The
cyclopentadiene anion is stabilized by five equivalent resonance structures.
The anion is an aromatic anion by virtue of it being a six-π -electron system. The indenyl anion is stabilized by a total of
seven resonance contributors. However, they are nonequivalent and all but one
require that the aromatic cloud of the benzene ring is disrupted. Thus, while
the negative charge is well delocalized, the resonance stabilization is less
than that of the cyclopentadiene system. Thus the proton is not as easily
removed, making indene a weaker acid.
15. It has been
found by NMR measurements that the α-methylene
groups (∗) of N -acetylpyrrolidine 1 are not
equivalent whereas the α-methylene
groups in N -(2-propenyl)-pyrrolidine
2 are equivalent. Provide an expla-nation based on resonance.

Answer:
If
the CH2 groups of 1 are
not equivalent, then the rate of rotation of the acetyl group is slow so that
one methylene group is in the vicinity of the C=O group and the other is in the
vicinity of the CH3 group. Such is not the case for 2. The rotation of the isopropenyl
group is rapid and the methylene groups experience an averaged environment.
Because of the electronegativity of oxygen, contributions of 1a to 1 are more important than the contributions of 2a to 2. Thus 1 has greater C–N double-bond character
and it is difficult to rotate about that bond. The C–N double-bond character is
small in 2, so rotation is facile,
leading to an averaged environment.

Related Topics
