Free-Radical Cyclization
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Carbon-Carbon Bond Formation By Free-Radical Reactions
With good methods available for producing carbon-centered free radicals, the cyclization process can be examined in greater detail. Cyclization involves the intramolecular addition of a free-radical to a double bond.
FREE-RADICAL CYCLIZATION
With
good methods available for producing carbon-centered free radicals, the
cyclization process can be examined in greater detail. Cyclization involves the
intramolecular addition of a free-radical to a double bond. Of course, this
requires that the two reacting parts of the molecule, the free-radical center
and the π bond, come within bonding
distance of one another.
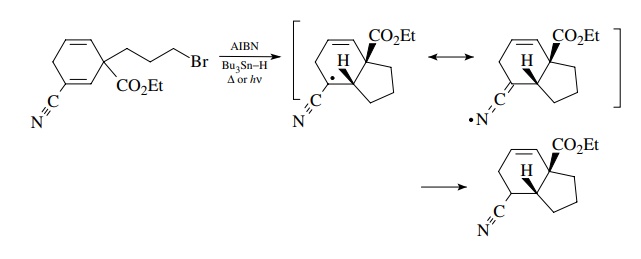
It
is quite easy for open-chain systems to undergo intramolecular cyclization
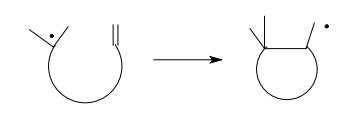
because of their many rotational degrees of freedom. More rigid systems undergo
efficient cyclization only if the free-radical center and the π system are held in close proximity, as
in the first example below. Where the molecular geometry is fixed in such a way
as to prevent effective interaction between the free-radical center and the π system, cyclization is inefficient and
reduction predominates. Cyclization in the second example is an obvious
impossibility!
Other
cases are not always so obvious, yet any structural or steric feature which
influences the close approach of the π
bond and the free-radical center will influ-ence the rate of cyclization and
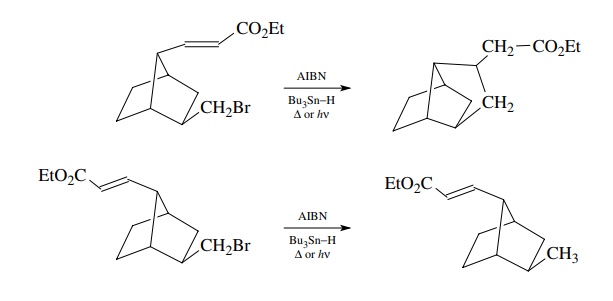
hence the yield of cyclized product. For example, trans-fused cyclopentyl
systems are much higher in energy than cis-fused ones; thus the trans-fused
cyclopentyl compound does not cyclize effectively and gives only reduction,
whereas
the cis-fused cyclizes efficiently
with little reduction,
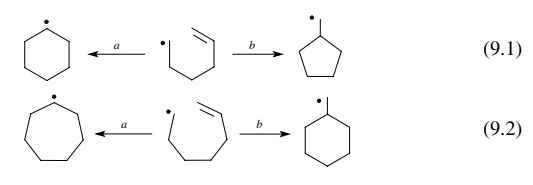
The
cyclization itself can produce two different ring sizes depending on which
carbon of the double bond is attacked. Of the two possibilities, it is seen
that one mode of cyclization gives a secondary radical while the other mode
produces a primary free radical.
Since
the order of free-radical stabilities falls in the order 3◦ > 2◦ > 1◦ , product stability
would dictate that cyclization should preferentially occur to give the more
stable secondary radical—a six-membered ring in reaction (9.1) (path a) and a seven-membered ring in reaction
(9.2)(path a).
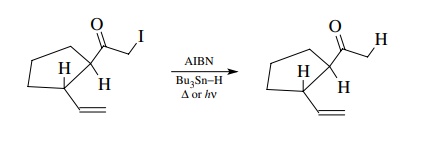
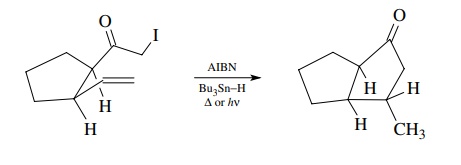
In
contrast, is known that the rates of ring-forming free-radical cyclizations are
5 > 6 > 7. Experimentally it was found that reaction (9.1) gives the
five-membered ring product (path b)
exclusively, and reaction (9.2) gives the six-membered ring product (path b). Thus the regioselectivity of ring
formation is controlled not by thermodynamic considerations but by kinetic
control of the cyclization. It turns out that bond formation between a radical
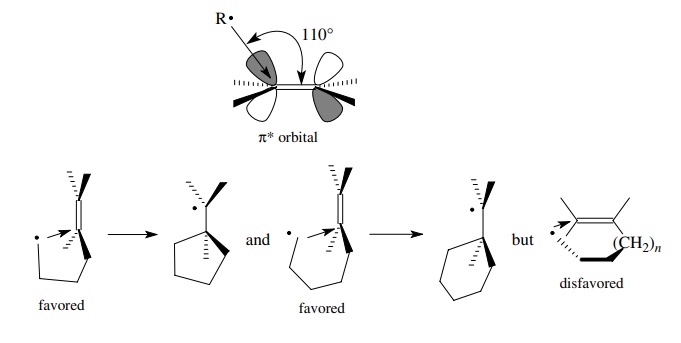
and a π system stereoelectronically
requires an approach angle of about 110◦ between the free-radical center and
the olefinic plane. (This is due to the fact that free-radical addition results
from donation of the unpaired electron on the radical into the π ∗ antibonding orbital
of the olefin, which coincidentally makes an angle of about 110◦ with the olefinic
plane.)
In
an intramolecular cyclization, attack on the end of the double bond closest to
the radical center (an exocyclic cyclization) achieves the proper approach
angle. Attack on the other olefinic carbon requires that the radical reach
across the double bond to achieve the proper approach angle. This is a higher
energy path and is kinetically disfavored. The same arguments hold for
cyclizations which can produce six- or seven-membered rings.
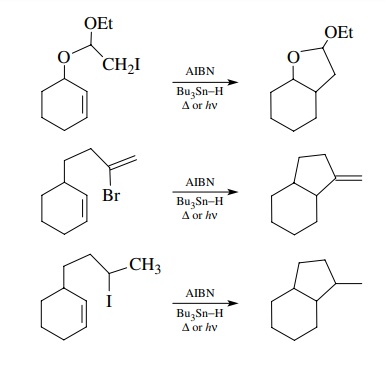
A
final feature of radical cyclizations is that they are mainly influenced by
steric factors and are practically insensitive to inductive effects. Since free
radicals are charge neutral, their reactivity is not greatly influenced by
either electron-donating or electron-withdrawing groups. For instance, the
following cyclizations occur with similar efficiencies even though the
electronic character of the cycliz-ing radicals are vastly different:
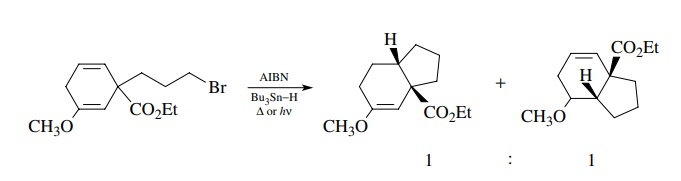
It
has also been shown that the electronic character of the olefin to which the
radical adds has little influence on the efficiency of the intramolecular
cyclization. Intramolecular competition between addition to an electron-rich
enol ether or a simple double bond gives a 1 : 1 ratio of products,
demonstrating that free-radical cyclizations have a remarkable insensitivity to
inductive effects.
Resonance
effects, on the other hand, can significantly affect the regiochem-istry of the
cyclization. Resonance delocalization of the unpaired electron of a free
radical stabilizes that radical. This is why the allyl radical is much more
stable than the n-propyl radical.
Thus, if a double bond is substituted with a group capable of providing
resonance stabilization to a free radical, it undergoes free-radical addition
much more readily than a double bond which cannot provide such resonance
stabilization.
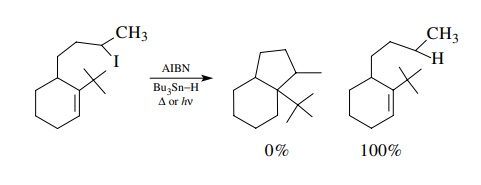
Steric
effects can also influence the cyclization process markedly. Bulky
sub-stituents which hinder the approach of the free radical to the π system can prevent cyclization
altogether and give only reduced product.
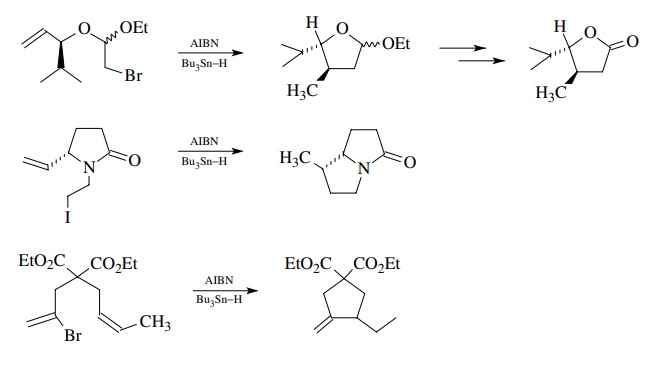
Below
are shown a few examples of the types of complex structures that can be
assembled by intramolecular free-radical cyclization. Note the presence of a
great many polar functional groups present in the cyclization substrates which
are compatible with the process. While the examples shown do not need
protecting groups, a great number of other free-radical cyclizations are known
which have unprotected alcohols, carbonyl groups, and carboxylic acids in the
cyclization precursor.
Free-radical
cyclization reactions nicely complement the Pd(0)-catalyzed in-tramolecular
Heck reaction, which also provides cyclic products from unsaturated halides.
Free radicals can be generated easily at saturated carbons from saturated alkyl
bromides, and the products are reduced relative to the reactants. In contrast,
intramolecular Heck reactions work best for vinyl and aryl bromides (in fact
they do not work for alkyl halides), and the products are at the same oxidation
level as the reactants. Moreover, free radicals attack the double bond at the
internal position, whereas palladium insertion causes cyclization to occur at
the external carbon.
The
advances made in using free radicals as synthetic intermediates in the last
10–20 years have been extraordinary due to new methods to effectively generate
free radicals and new insights into their reactivity patterns which allow them
to be controlled. As a consequence, the construction of ring systems has been
tremendously facilitated.
Related Topics
