Formation of Enantiomers
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Stereochemical and Conformational Isomerism
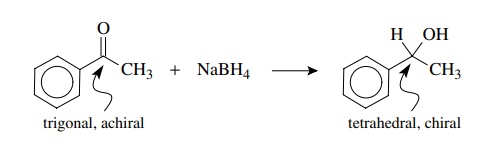
Since chiral centers are most commonly tetrahedral, the conversion of trigo-nal centers to tetrahedral centers by some type of addition process is the most common way in which new chiral centers are created.
FORMATION OF ENANTIOMERS
Since
chiral centers are most commonly tetrahedral, the conversion of trigo-nal
centers to tetrahedral centers by some type of addition process is the most
common way in which new chiral centers are created. The reaction of carbonyl
groups with nucleophiles is a classic example. If substituents on the carbonyl
group and the nucleophile are all different, then a new chiral center is
pro-duced, as in the reaction of acetophenone with sodium borohydride to
produce 1-phenylethanol.
The
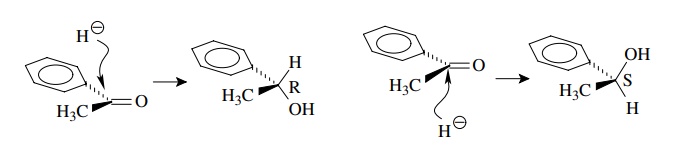
carbonyl group is trigonal and planar and can be thought of as having two
faces. Addition of hydride to one face gives one enantiomer while addition to
the opposite face gives the opposite enantiomer. As rewritten below, attack
from above gives the R enantiomer while attack from below gives the S
enantiomer. The faces are stereochemically nonequivalent since different
stereoiso-mers are produced.
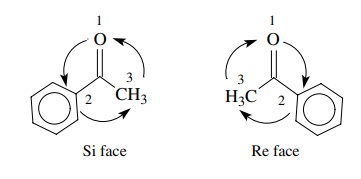
To
differentiate the faces of a carbonyl group, the Re – Si nomenclature has been
developed. The groups around the carbonyl carbon are given priorities by the
same rules used in the Cahn – Ingold – Prelog system for R,S nomenclature. Then
going from the group of highest priority to the group of lowest priority around
the face of a carbonyl group, proceeding in the clockwise direction defines the
Re face and proceeding in the counterclockwise direction defines the Si face.
The
Re – Si nomenclature enables the faces of a carbonyl group to be
differen-tiated stereochemically; however, the carbonyl group itself is
achiral. Moreover, the Re – Si designation is not indicative of the
stereochemistry of the chiral center produced by addition. In the above example
hydride addition to the Si face gives the R enantiomer while hydride addition
to the Re face gives the S enantiomer. If ethyl lithium were added, the
stereochemistry would be reversed, that is, Si → S and Re → R.
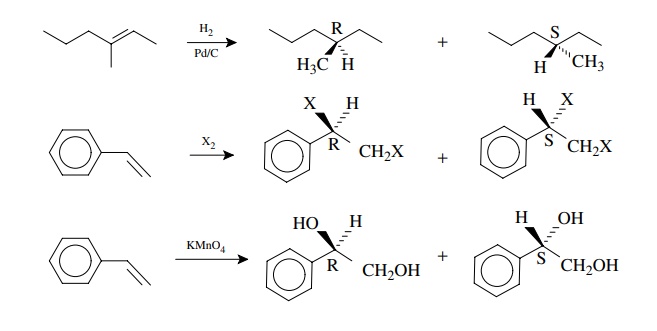
New
chiral centers are produced by addition reactions to other trigonal centers as
well. Hydrogenation of 3-methyl-3-hexene gives 3-methylhexane. Clearly the
addition of hydrogen to one face of the planar olefinic system gives one
enan-tiomer and addition to the opposite face gives the opposite enantiomer.
Likewise reaction of styrene with chlorine or bromine (X2) or
potassium permanganate produces products with a new chiral center. Formation of
the two possible enan-tiomers results from addition to either face of the
olefin.
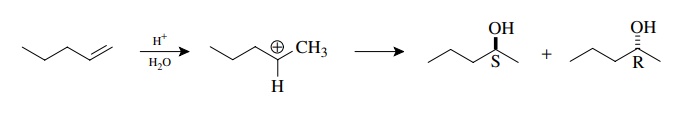
Reactive
intermediates which are planar can also produce enantiomers. The acid-catalyzed
addition of water to 1-pentene proceeds via a secondary carboca-tion. Because
the carbocation is a trigonal, planar intermediate, water can add to either
face to give the R or S enantiomers.
In
reactions in which neither the reactants (C=O, C =C, C+) nor the reagents
(BH4−,
EtMgBr, Br2, H2O, etc.) are chiral, there is no possibility
for controlling which face undergoes addition (in fact, addition to either face
is equivalent); thus a racemic mixture will be produced. Such processes are
described as having no enantioselectivity.
Related Topics
