Fixed Oils And Fats
| Home | | Pharmacognosy |Chapter: Pharmacognosy and Phytochemistry : Drugs Containing Lipids
Fixed oils and fats are obtained from plants or animal. They are rich in calories and in plant source, they are present mostly in the seeds, as reserve substances and in animals they are present in subcutaneous and retroperitoneal tissues.
FIXED OILS AND FATS
Fixed oils and fats are obtained from plants or animal. They
are rich in calories and in plant source, they are present mostly in the seeds,
as reserve substances and in animals they are present in subcutaneous and
retroperitoneal tissues. They differ only according to their melting point and
chemically they belong to the same group. If a substance is liquid at
15.5–16.5°C it is called fixed oil and solid or semisolid at the above temperature,
it is called fat. They are made from two kinds of molecules: glycerol (a type
of alcohol with a hydroxyl group on each of its three carbons) and three fatty
acids joined by dehydration synthesis. Since there are three fatty acids
attached, these are known as triglycerides. These fatty acids may be saturated,
monounsaturated or polyunsaturated. The terms saturated, mono-unsaturated, and
poly-unsaturated refer to the number of hydrogens attached to the hydrocarbon
tails of the fatty acids as compared to the number of double bonds between carbon
atoms in the tail. Fats, which are mostly from animal sources, have all single
bonds between the carbons in their fatty acid tails, thus all the carbons are
also bonded to the maximum number of hydrogens pos-sible. Since the fatty acids
in these triglycerides contain the maximum possible amount of hydrogens, these
would be called saturated fats. The hydrocarbon chains in these fatty acids
are, thus, fairly straight and can pack closely together, making these fats solid
at room temperature. Oils, mostly from plant sources, have some double bonds
between some of the carbons in the hydrocarbon tail, causing bends or ‘kinks’
in the shape of the molecules. Because some of the carbons share double bonds,
they are not bonded to as many hydrogens as they could if they weren’t double
bonded to each other. Therefore these oils are called unsaturated fats. Because
of the kinks in the hydrocarbon tails, unsaturated fats can’t pack as closely
together, making them liquid at room temperature.
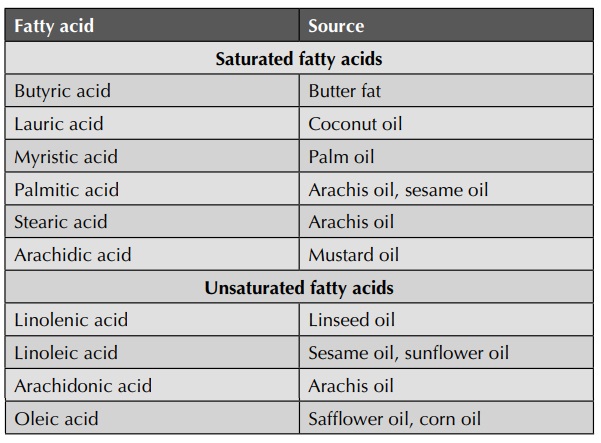
Examples of saturated and unsaturated fatty acids are given
in table below.
Table : Examples of saturated and unsaturated fatty acids
Fixed oils and fats are insoluble in water and alcohol and
are soluble in lipid solvents like light petroleum, ether, chloroform, and
benzene. Only exception in this solubility is castor oil that is soluble in
alcohol because of its hydroxy group of ricinoleic acid. They float in water
since their specific gravity is less than one. They produce a permanent
translucent stain on the paper and are called fixed oils. Fixed oils and fats
cannot be distilled without their decomposition.
Analytical Parameters for Fats and Oils
Following are the parameters used to analyse the fats and
oils.
1) Iodine
value: The iodine
value is the mass of iodine in grams that is consumed by 100 g of fats or oil. A iodine
solution is violet in colour and any chemical group in the substance that
reacts with iodine will make the colour disappear at a precise concentration.
The amount of iodine solution thus required to keep the solution violet is a
measure of the amount of iodine sensitive reactive groups. It is a measure of
the extent of unsaturation and higher the iodine value, the more chance for
rancidity.
2) Saponification
value: The
saponification value is the number of milligrams of potassium hydroxide required to
saponify 1 g of fat under the conditions specified. It is a measure of the
average molecular weight of all the fatty acids present.
3) Hydroxyl
value: The hydroxyl
value is the number of mg of potassium hydroxide (KOH) required to neutralize
acetic acid combined to hydroxyl groups, when 1 g of a sample is acetylated.
4) Ester value: The ester value is the number
of mg of potassium hydroxide (KOH) required to saponify the ester contained in
1 g of a sample.
5) Unsaponifiable
matter: The
principle is the saponification of the fat or oil by boiling under reflux with
an ethanolic potassium hydroxide solution. Unsaponifiable matter is then
extracted from the soap solution by diethyl ether. The solvent is evaporated
and then the residue is dried and weighed.
6) Acid
value: It is the
amount of free acid present in fat as measured by the milligrams of potassium hydroxide
needed to neutralize it. As the glycerides in fat slowly-decompose the acid
value increases.
7) Peroxide
value: One of the
most widely used tests for oxidative rancidity; peroxide value is a measure of the
concentration of peroxides and hydroperoxides formed in the initial stages of
lipid oxidation. Milliequivalents of peroxide per kg of fat are measured by
titration with iodide ion. Peroxide values are not static and care must be
taken in handling and testing samples. It is difficult to provide a specific
guideline relating peroxide value to rancidity. High peroxide values are a
definite indication of a rancid fat, but moderate values may be the result of
depletion of peroxides after reaching high concentrations.
