Enzymes and Enzyme Characteristics
| Home | | Anatomy and Physiology | | Anatomy and Physiology Health Education (APHE) |Chapter: Anatomy and Physiology for Health Professionals: Levels of Organization : Chemical Basics of Life
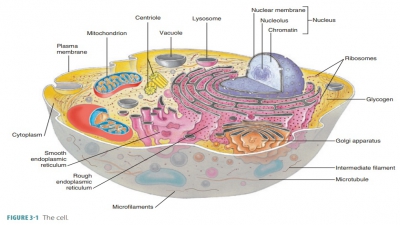
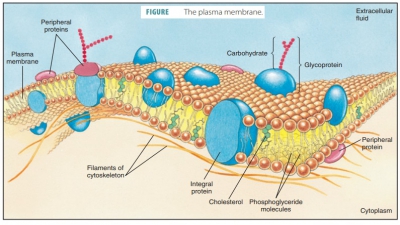
Enzymes are globular proteins that promote chemical reactions by lowering the activation energy requirements.
Enzymes
Enzymes are globular proteins that promote chemical reactions by
lowering the activation
energy requirements.
Activation energy is the energy that must be overcome for a chemical reaction
to occur. Therefore, they make chemical reactions possible and catalyze the
reactions that sustain life. This means that enzymes are catalysts. Enzyme
molecules are manufactured by cells to promote specific reac-tions. Enzymes are
among the most important of all the body’s proteins. Nearly everything that
occurs in the human body relies on a specific enzyme. In the body, enzymes
assist in the digestion of food, drug metabolism, protein formation, and many other
types of reactions. Enzymes make metabolic reac-tions possible inside cells by
controlling tempera-ture conditions that otherwise would be too mild for them
to occur.
Enzymes are complex molecules.
When they are not used in the reactions they catalyze, they are recy-cled.
Enzymatic reactions, which are reversible, can be written as:
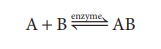
Enzymes
cannot cause a chemical reaction between molecules that would not react without
them.
They increase the speed of enzymatic reactions greatly, between
100,000 to more than 1 billion times the rate of a reaction that is
uncatalyzed. Otherwise, biochemical reactions would occur extremely slowly,
almost to no effect. Enzymes are vital in making these reactions occur at an adequate
pace.
Enzyme Characteristics
Enzymes differ in their makeup.
Some are only made of proteins, whereas others have a two -part struc-ture,
consisting of a protein portion (the apoenzyme)
and a cofactor. Collectively, these two parts are referred to as the holoenzyme. Enzyme
cofactors may be either a metal element ion (such as iron or copper) or an
organic molecule that assists the reac-tion. Most organic cofactors are derived
from B (or other) vitamins, in which case they are referred to as coenzymes.
Enzymes have chemical-specific
actions. Some control one chemical reaction, whereas others regu-late a small
group of similar reactions by binding to molecules that are only slightly
different. Enzymes act on substances referred to as substrates. Certain enzymes, when present, determine which reactions are sped up
and which reactions will occur. If there is no enzyme, there is no reaction.
Enzymes are often named after their substrates, using the suffix -ase. For example, a lipid is catalyzed
by an enzyme called a lipase. Another
enzyme, called a catalase, breaks
downhydrogen peroxide into water and oxygen. Hydrogen peroxide is a toxic
substance that results from certain metabolic reactions.
Every cell holds hundreds of
various enzymes, each of which recognizes its specific substrates. Enzyme
molecules have three-dimensional shapes (conformations) that allow them to
identify their substrates. The coiled and twisted polypeptide chain of each
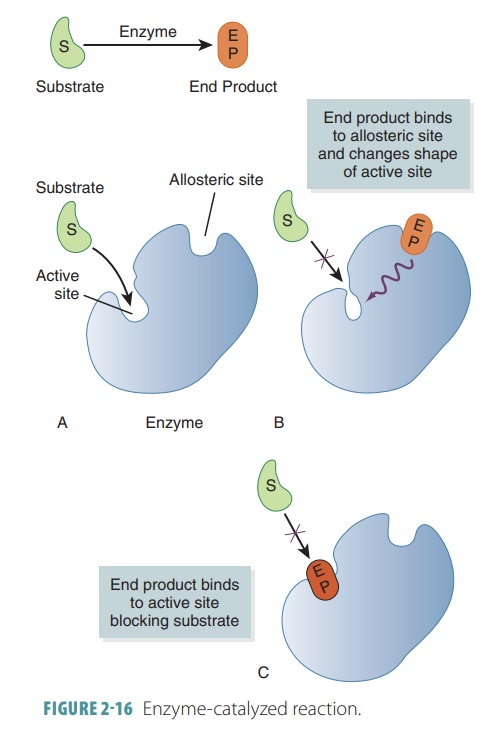
enzyme fits the shape of its substrate. The activesite
of an enzyme molecule combines with
portionsof its substrate molecules temporarily. This forms an enzyme–substrate
complex (FIGURE 2-16).
When enzyme–substrate complexes
are formed, some chemical bonds within the substrates are distorted or
strained. Requiring less energy as a result, the enzyme is released as it was
originally configured. Enzyme-catalyzed reactions can be summarized as:

These reactions are often
reversible. Sometimes, the same enzyme catalyzes the reaction in both
directions. The reactions occur at differing rates, based on the number of
molecules of the enzyme and its substrate. Some enzymes process a few substrate
molecules every second, whereas others can process thousands in the same
length of time.
1. Define
enzymes and explain their functions.
2. Define
the terms cofactor, holoenzyme, and coenzymes.
3. Define
substrates.