Design of Lead and Lead Discovery
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Concepts of Drug Design
The concept of lead discovery envisages two investigational processes. They are: 1. Exploration of leads 2. Exploitation of leads
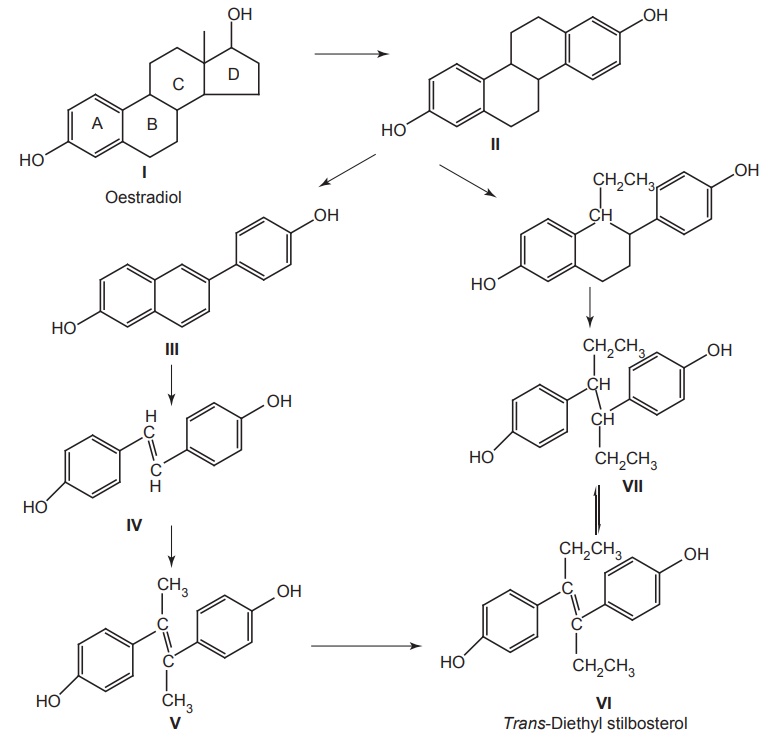
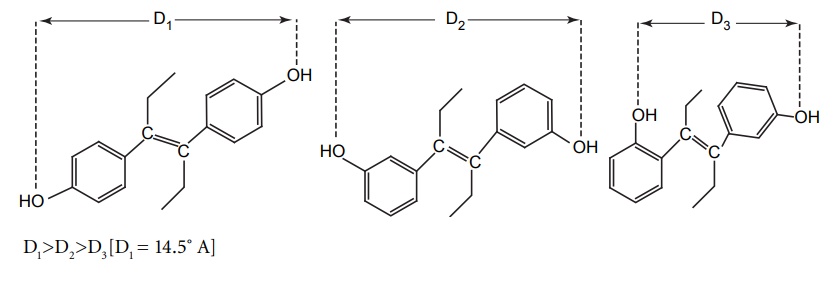
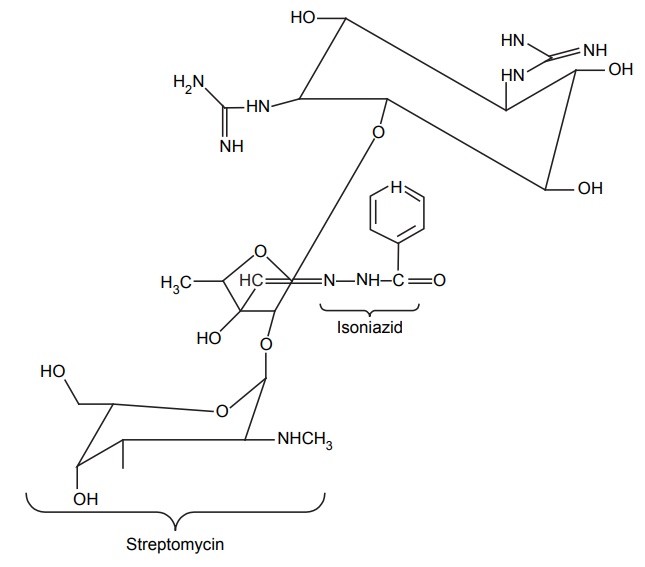
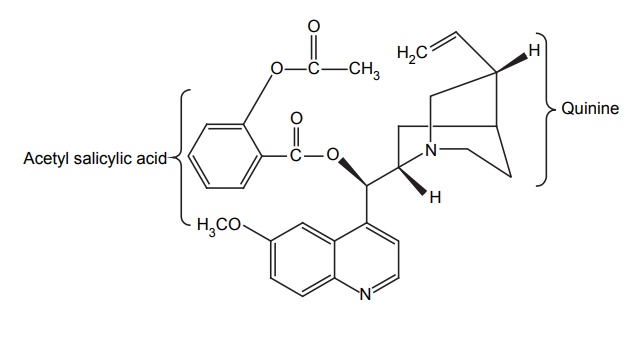
DESIGN OF LEAD AND LEAD DISCOVERY The concept of lead discovery envisages two investigational processes. They are: Exploration of leads: search of new molecules. Exploitation of leads: assessment, chemical modelling, and extension of leads. The drug discoveries without a lead are quiet few in number. The most prominent examples include penicillium and librium. Librium is the first benzodiazepine tranquillizer. A series of quinazoline-3-oxides were synthesized by Leo Sternbach at Roche in a new tranquilizer development programme. None of the molecules produced reliable pharmacological activity. One of the molecules that had been missed and identified while a laboratory clean up was happening, found to posses probable activity and chemically it was benzodiazepine-4-oxide. It produced chloromethyl quinazoline-3-oxide with methylamine. This lead identification was further exploited to develop potent analogues such as diazepam, which was found to be 10 times more potent than the lead. The lead identifications require a series of biological evaluation of the lead molecules. Once, after the identification, it can be structurally modified, the potency and the activity are improved. The entire synthesized compounds or any chemical constituents obtained from natural products are evaluated in a series for their biologically active components. Thus, random screening may produce unexpected active medicines. Antibiotics, such as streptomycin, tetramycins, and fungal metabolites, such as lovastatin and cyclosporins, were found through this method. This approach needs more manpower, and it is expensive and time-consuming and the success rate is considerably low. In this method, only compounds that possess similar structural skeletons were evaluated from their particular properties. PHARMACOKINETIC STUDIES Biotransformation occurs as the fate by metabolizing enzymes. In order to develop new leads, the metabolites or biotransformed compounds are studied for their properties, and such studies are expected to asses the activity from a comparison with the parent molecule. For example, the discovery of sulphanilamide is reported through the metabolic studies of prontosil. PHARMACODYNAMIC STUDIES The effects apart from the therapeutic actions, that is, side effects may lead to the finding out of a new molecule with some appreciable structural modification. For example, sulphonamide used specifically for the treatment of typhoid, lowered the blood sugar levels drastically. This exerted action led to the finding of aryl sulphonyl thiourea moiety responsible for the lowering of blood glucose level. Amino alkyl derivatives of iminodibenzyl were synthesized as analgesic, sedative, and antihistamines that was found to posses antidepressive action. This lead to the synthesis of many tricyclic antidepressants. Disjunction comes into the systemic formulation of analogues of a prototype agent, generally, towards structurally simpler products, which may be carried as quasi-replicas or partials of the prototype agent. This is employed by various methods, that is Unjoining of certain bonds. Substitution of aromatic cyclic system for saturated bonds. Elimination of the size of hydrocarbon portion of the parent molecule. Decyclization of any ring system. Cyclization of hydrocarbon chains. For example, oestrogenic study of oestradiol through drug design by disjunction produced successful molecules of trans-diethylstilbosterol. The disjunction of various steps in the design from II to III and to IV has not successively produced a reliable molecule, but it has succeeded in the total elimination of ring B and C in estradiol (I). By plotting the response curve, the maximal activity in the series was attributed to trans-diethylstilbosterol (D1) and the possibility of reductions in activity depends on the distance between two hydroxyl groups. In this method, a systemic formulation of analogues of a prototype agent is employed. In general, principally mixed moieties are derived by conjunction of two pharmacophoreic molecules. An example for this is ganglionic blocking agents and its development is based on the principle of mixed moieties. Acetylcholine is a neurotransmitter, which acts as a parasympathetic muscarnic stimulant and produces appreciable changes in ganglionic functions; whereas, hexamethonium is a ganglionic blocker, and posses only a slight action at postganglionic parasympathetic endings and produces a high degree of ganglionic blockade. The evaluation of Muscarnic moiety on being studied in relation with a particular bisquartenary type of structure, for example, hexamethonium, promptly suggests the following proposed design, thus, embodying the ganglionic moiety and Muscarnic moiety into a single molecule. It is, however, pertinent to mention here that the internitrogen distance essentially constitute an important factor in many of the series of bis quarternary salts possessing ganglionic blocking activity. It is worthwhile when the distance is more or less the same as that in hexamethonium. However, the actual synthesis and pharmacological evaluation of conjunctioned hexamethyl analogue reveals the presence of both a weak Muscranic stimulant and possessing of a good ganglionic blocking action. Molecular hybridization essentially embodies the synthesis of strategically designed new breeds of bioactive agents from two or more compounds having different characteristic features with the aid of covalent-bond synthesis. In 1886, Necki exploited the beneficial properties of phenols and carboxylic acids possessing potent antibacterial characteristic feature into the design of newer drug molecules with better and improved pharmacological activities by simple esterification. Example 1. A molecule of streptomycin and a molecule of isoniazid by means of a strong double bond between ‘C’ and ‘N’ forms a hybridized molecule through the elimination of a molecule of water. This hybridized molecule exhibits significant potentiated antibacterial and tuberculostatic activity. 2. Hybridization of acetyl salicylic acid (antipyretic) and quinine (antimalarial) to lose a molecule of water to form a hybridized molecule for a potent antimalarial drug with substantial antipyretic and analgesic activity.Approaches to Lead Discovery
RANDOM SCREENING
NONRANDOM SCREENING
Drug Design Through Disjunction
Drug Design Through Conjunction
MOLECULAR HYBRIDIZATION IN DRUG DESIGN
Related Topics
