Background
| Home | | Hospital pharmacy |Chapter: Hospital pharmacy : Purchasing medicines
The pharmaceutical market in the UK forms the context for hospital purchasing.
Background
Expenditure
The pharmaceutical
market in the UK forms the context for hospital purchasing. The UK represents
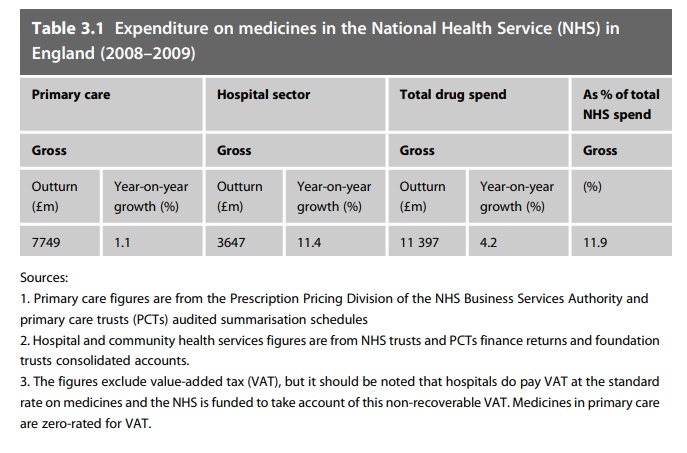
around 3% of the global market for medicines. NHS hospital expenditure, at just
over £3.6 billion, represents about a third of the NHS total for prescription
medicines, with the balance being spent in primary care (Table 3.1). This sum
excludes value-added tax (VAT: a purchase tax), but it should be noted that
hospitals do pay VAT on medicines (where applic-able – some items do not incur VAT)
and this element is included in the expenditure figures – figures for general
practitioner prescribing costs do not include VAT. Overall expenditure on
medicines at over £11 billion forms around 12% of NHS expenditure.
The Pharmaceutical Price Regulation Scheme (PPRS)
The PPRS is a UK government scheme that controls the prices of branded prescription medicines supplied to the NHS by regulating the profits that pharmaceutical companies can make on their sales to the NHS. At the same time, the scheme supports an industry that will continue to offer innovative medicines and is competitive internationally. Indeed, the UK pharmaceutical industry is seen as a significant asset; The NHS Plan made this clear and also stated there needs to be opportunity for companies to undertake research with reasonable haste.
Under the PPRS
suppliers are allowed to introduce major new medicines (new active substances)
to the NHS at prices that they determine but can only increase prices with the
Department of Health’s agreement. The PPRS distin-guishes the UK market from
all others in the European Union (EU), though at the time of writing a
value-based pricing approach is being considered.
Generic substitution
Unlike the current
system in primary care, NHS hospital pharmacies have dispensed medicines
generically, irrespective of how they are prescribed, for some time. Thus, a
brand name may be used by the doctor but a generic version issued. Exceptions
are made if the branded product has unique characteristics that could result in
a clinically important effect if substituted (modified release, for example).
Related Topics
