Adverse drug reactions
| Home | | Hospital pharmacy |Chapter: Hospital pharmacy : Risks with medicines
When a medicine is first licensed and marketed there will be limited information about its side-effects and potential to cause adverse drug reactions.
Adverse drug reactions
When a medicine is
first licensed and marketed there will be limited information about its side-effects
and potential to cause adverse drug reactions. For a medicine to be safe the
benefits to the patient of taking the medicine should be greater than the risks
of harm to the patient. The marketing authorisation for the drug is granted on
the basis of a balance of benefits and risks. Once a drug is used more widely
and more doses are taken by patients, more side-effects will become evident,
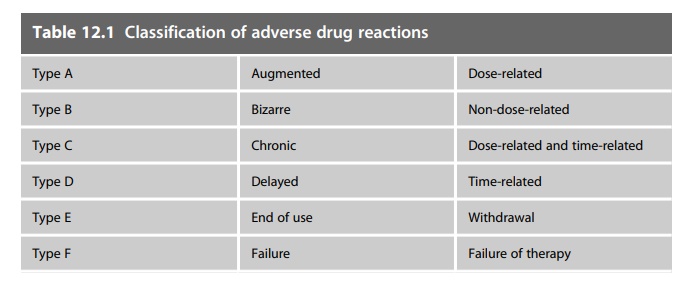
and less common and potentially more serious side-effects will emerge. Table
12.1 provides a classification of adverse drug reactions.
To protect patients
from the risks of medicines a system must be in place to be able to collect
information about, and identify trends in, the adverse drug reactions these
medicines may cause. Pharmacovigilance is a process for monitoring the use of
medicines once they have been licensed for use to ensure information about
adverse effects not identified prior to the drug being marketed can be
collected and collated. It is also a means of identi-fying changes in the
patterns of adverse effects to drugs already widely used. By understanding
these patterns and the frequency of adverse effects, risks and benefits can be
assessed to determine whether action needs to be taken to improve their safety,
for example, by providing additional infor-mation to prescribers and patients
about cautions and contraindications. The ultimate aim of pharmacovigilance is
to ensure medicines have a positive impact on patients and the risk of harm
from those medicines is minimised.
In the UK, the
Medicines and Healthcare Products Regulatory Agency (MHRA) is responsible for
monitoring medicines safety. It does this by col-lecting data about the use of
medicines from a wide variety of sources, includ-ing clinical studies,
published medical literature, pharmaceutical companies and from individual case
reports submitted to the MHRA as part of a spon-taneous reporting system, the
Yellow Card scheme.
The Yellow Card
scheme is a mechanism by which patients and healthcare professionals can report
actual and suspected adverse drug reactions to new and established medicines.
The reports are evaluated alongside other infor-mation to identify whether any
action needs to be taken to minimise the risk and maximise the benefits of the
drug to the patient by changing product information, restricting the
indications for using the medicine or, in extreme situations, removing the
product from the market completely.
Related Topics
