Preparation of Bacterial Specimens for Light Microscopy
| Home | | Pharmaceutical Microbiology | | Pharmaceutical Microbiology |Chapter: Pharmaceutical Microbiology : Identification of Microorganisms
As a large segment of living microorganisms invariably appear almost colourless when seen through a standard light microscope, one should always subject them to a highly specific treatment for possible vivid observation.
Preparation
of Bacterial Specimens for Light Microscopy
As a
large segment of living microorganisms invariably appear almost colourless when seen through a standard light microscope, one should always subject them to a
highly specific treatment for possible vivid observation. Staining (or colouring)
is regarded to be one of the widely accepted phe-nomenon to accomplish the
aforesaid objective.
The
various aspects of ‘staining’ shall
be duly elaborated in the following sequential manner, namely :
·
Stained preparations
·
Preparation of smears for staining
·
Gram staining
·
Differential staining
·
Miscellaneous staining e.g.,
Capsule
staining ; Endospore staining ; Flagellar staining.
Stained Preparations
In usual
practice a large number of investigative studies related to the specific shapes
and cellular arrangements of various microbes are effectively carried out with
the help of stained preparations. In
other words, different means and ways to colour the microorganisms with a
particular and appropriate dye (i.e.,
staining) is performed meticulously
so as to emphasize certain structures vividly and explicitely. It may be
worthwhile to state here that before one commences the ‘staining’ of the microbes they should be duly fixed (or attached)
onto the surface of the microscopic
slide ; naturally without proper fixing,
the requisite stain could wash them off the slide instantly.
Preparation of Smears for Staining
The ‘fixing’ of specific specimen may be
accomplished by first spreading a resonably thin film of the material onto the
surface of the microscopic slide. In fact, this ‘thin film’, is termed as smear,
which is subsequently air dried. The air dried slide is now carefully exposed
to a low flame of a Bünsen burner a number of times, taking special care that
the smear side is always up. The
aforesaid most common ‘staining
methodology’ comprising of air-drying
followed by flame-heating allows the
fixing of the microorganisms onto
the surface of the slide, and invariably kill
them completely. After this, the ‘suitable
stain’ is adequately applied, and subsequently washed off with ample
slow-running water. The wet slide is
now gently blotted with absorbent paper. The resulting slide having the stained microorganisms are actually ready for detailed microscopic examinations, whatsoever.
Gram Staining
Hans
Christian Gram (1884) – a Danish bacteriologist first and foremost developed
the well known staining procedure called as Gram staining. Since, its inception earned a well-deserved
recognition across the globe by virtue of the fact that it categorically
divides microorganisms into two major
categories, namely : (a) Gram-positive*, and (b) Gram-negative**.
Methodology : The various steps involved are as
follows :
(1) A heat-fixed bacterial smear is duly
covered with the following staining reagents in a sequential manner, namely : (a) crystal
violet (i.e., a basic purple dye) which eventually
imparts its colour to all cells ;
and hence usually referred to as a primary
strain ; (b) iodine solution i.e.,
clearly washing off the purple dye after a short while, the smear is covered
with iodine solution that serves as a mordant***
; (c) alcohol**** i.e., the
iodine is washed off thereby causing a
‘decolourizing effect’ ; and (d) safranin
– a basic red due (or other appropriate agent) i.e., to act as a counterstrain.
(2) The
resulting ‘smear’ is washed again,
blotted dry, and carefully examined microscopically.
(3) In
this manner, the purple dye (crystal violet) and the iodine combine with each bacterium thereby imparting to it
a distinct purple or dark violet colouration.
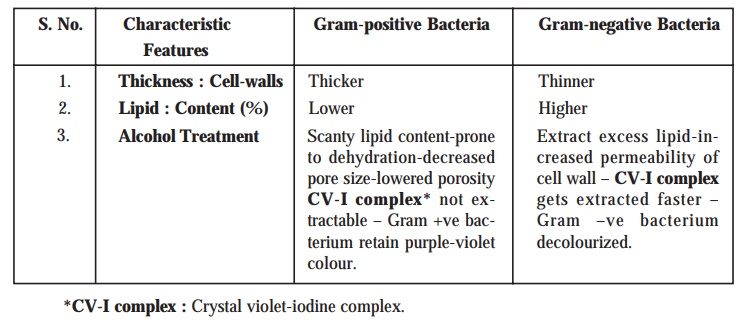
Gram-positive Bacteria : The
bacteria which ultimately retain the purple or dark violet colouration even after the alcohol treatment to
decolourize them are grouped together as Gram-positive
bacteria. Besides, it has been duly observed that as these specific class
of microorganisms do retain the original purple stain, they are significantly
not affected by the safranin
counterstain at all.
Gram-negative Bacteria : The
bacteria that eventually lose the crystal violet, are duly counterstained by the safranin
; and, therefore appear red in colour.
The
characteristic features enumerated below for Gram +ve and Gram –ve
bacteria vividly justifies why the Gram-staining
technique renders some microorganisms purple-violet and others red in
appearance.
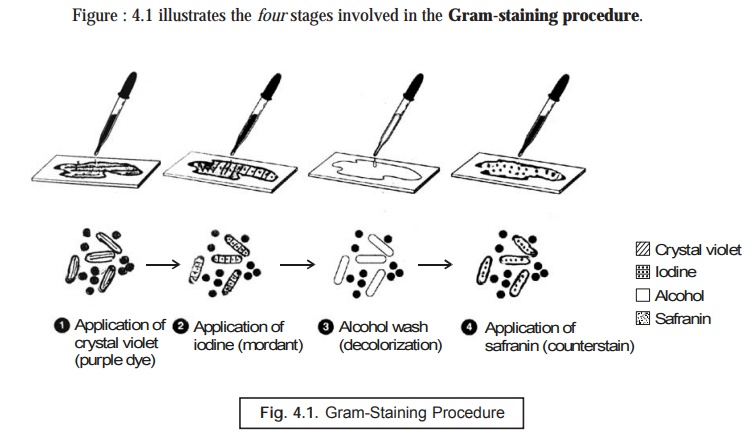
(1) A
heat-fixed bacterial smear of cocci and rods is first duly covered with a basic
purple dye (primary stain) e.g.,
crystal violet, and the dye is washed off subsequently.
(2) Resulting
smear is covered with iodine (a mordant), and washed off. At this particular
stage both Gram +ve and Gram –ve bacteria are purple in appearance.
(3) The
treated slide is washed with ethanol or an alcohol-acetone solution (a
decolourizer), and washed with water subsequently. At this stage Gram +ve cells
are purple, and Gram –ve cells are colourless.
(4) Final
step, safranin, is added as a counterstain, and the slide is washed, dried, and
examined micro-scopically. Gram +ve bacteria retain the purple dye, whereas the
Gram –ve bateria appear as pink.
[Adapted
from : Tortora et. al. Microbiology an Introduction, The
Benjamin/Cummings Publishing Co. Inc., New York, 5th edn, 1995].
Differential Staining
In bacteriology, a stain for instance Gram’s stain which evidently enables
one to differentiate distinctly amongst the various kinds of bacteria. It may
be emphasized at this material time that unlike simple stains, the differential stains very much interact altogether in a different manner with
specifically different types of
microorganisms ; and, therefore, this criterion may be exploited to afford a
clear cut distinction amongst them. In actual practice, however, the
differential stains largely employed for microorganisms are (a) the Gram’s stain ; and (b)
the Acid-Fast Stain.
Gram’s Stain
It has
already been discussed at length in the later Section.
Acid-Fast Stain
Acid- fast stain is used invariably in bacteriology, especially for staining Mycobacterium
tuberculosis, and
Mycobacterium leprae. This acid-fast stain possesses an inherent ability to get bound intimately only to such
microbes that have a waxy material in their cells (e.g., all bacteria in the genus Mycobacterium).
Besides, this particular stain is also employed to identify precisely the
disease-producing stains belonging to
the genus Nocardia.
Methodology : The various steps involved in the acid-fast stain are as enumerated
under :
(1) A
specially prepared solution of the red dye carbolfuschin
is generously applied onto the exposed surface of a heat-fixed bacterial smear
; and the treated slide is warmed* gently for several minutes.
(2) The
slide is brought to the room temperature (cooled) and washed duly with water.
(3) The,
resulting smear is now treated with acidic-alcohol
(i.e., a decolourizer) that removes critically the red stain from
microorganisms which are not acid-fast.
(4) Thus,
the acid-fast microbes do retain the
red colour (due to carbolfuschin) by
virtue of the fact that the red dye shows far greater solubility in the waxes present in the cell wall rather
than the acid-alcohol.
(5) In non-acid-fast microorganisms, whose
cell walls are devoid of specific waxy compo-nents, the dye carbolfuschin gets readily removed in
the course of decolourization
thereby rendering the cells almost colourless.
(6) Finally,
the resulting smear is duly stained with methylene
blue counterstain whereby the non-acid-fast
cells appear blue distinctly and
the acid-fast cells as red.
Ziehl-Neelsen Method (for
staining M. tuberculosis) : This method was developed by two noted scientists, namely : (a) Franz
Ziehl – a German Bacteriologist
(1857-1926), and (b) Fried rich Karl Adolf Neelsen – a German Pathologist (1854-1894), whereby the causative organism M. tuberculosis could be stained effectively. A
solution of carbolfuschin is applied
duly, which the organism retains after usual rinsing with acid-alcohol admixture.
Miscellaneous Staining
There are
centain equally important staining procedures which do not fall within the
techniques discussed under Sections 4.6.1.1. through 4.6.1.4. Hence, these
special staining procedures shall be treated individually in the sections that
follows :
Capsule Staining (or Negative Staining for Capsules)
Capsule : The bacterial capsule refers to the membrane that particularly
surrounds certain bacterial cells,
thereby offering adequate protection against the phagocytosis* and allowing evasion of host-defense mechanisms**.
It has
been duly observed that a host of microorganisms essentially comprise of a
gelatinous covering (i.e., capsule). However, in the domain of medical microbiology the very presence
of a capsule specifically
establishes the virulence*** of the
said organism, the extent to which a pathogen may be able to cause disease.
In
general, the capsule staining is
rather more complicated and difficult in comparison to other kinds of staining techniques due to the fact
that the particular capsular materials
are not only water soluble but also removable during the thorough washing
procedure.
Methodology : The various steps involved during
the capsule staining are as stated
under :
(1) First
of all the microorganisms are carefully mixed in a solution comprising of a fine colloi-dal suspension of some
distinct coloured particles (one may
invariably make use of either nigrosin or India ink) to afford a dark
background.
(2) The
bacteria may now be stained duly with a simple
stain, for instance : safranin.
(3) By
virtue of the fact that capsules do
have a highly peculiar chemical
composition fail to accept a plethora of ‘biological dyes’ e.g., safranin ; and, therefore, they mostly
appear as haloes just
surrounding every stained microbial cell.
(4) Importantly,
the application of India ink duly
demonstrates a negative-staining
procedure so as to give rise to a distinct contrast between the capsule and the adjoining dark medium.
Endospore (Spore) Staining
Endospore refers to a thick-walled spore
produced by a bacterium to enable it to survive unfavourable environmental conditions. In actual practice, the
occurrence of endospores are
comparatively not-so-common in the microbial cells ; however, they may be
adequately generated by several genera of microorganisms. It is pertinent to
mention here that the endospores
cannot be stained by such ordinary techniques as : (a) simple staining ; and
(b) Gram staining, due to the fact the biological dyes are incapable of penetration through the wall of
the endospore.
Schaeffer-Fulton Endospore Stain (or
Schaeffer-Fulton Procedure) : In the Schaeffer-Fulton procedure the
endospores are first and foremost stained by heating together the
respective microorganisms with malachite green, that happens to be a very strong stain which is capable of
penetrating the endospores. Once the
malachite green treatment is duly
carried out, the rest of the cell is washed rigorously free of dye with water,
and finally counterstained with safranin.
Interestingly, this specific technique gives rise to a green endospore clearly resting in a pink to red cell as depicted in Fig. 4.2.
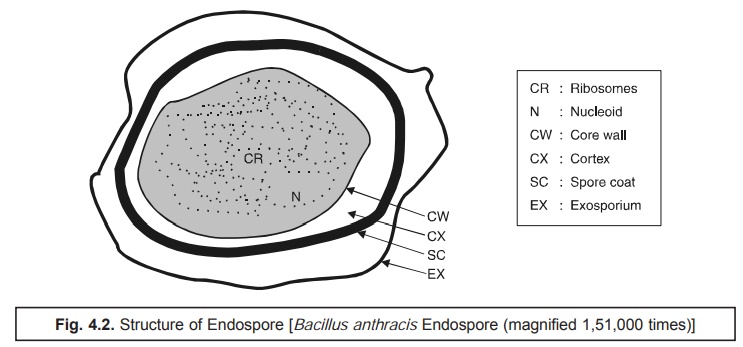
As the endospores are highly refractive in nature, they may be visualized explicitely (i.e., detected) under the light microscope when unstained*.
Flagella Staining
Flagella (Pl. of Flagellum) usually refers to a threadlike structure that
essentially provides motility for
certain microorganisms and protozoa (one, few, or many per cell),
and for spermatozoa (one per cell).
It has
been well established that the bacterial
flagella do represent various structures of locomotion that happen to be
exceptionally small to be visualized with the help of light microscopes without staining.
Methodology : The staining technique consists
of a tedious and a quite delicate stepwise procedure that makes use of a stain carbolfuschin
and a mordant so as to build up the
desired requisite diameters of the respective flagella unless and until they
are rendered quite reasonably visible under the light microscope. Clinical
microbiologists usually exploit the arrangement and the specific number of flagella critically
as diagnostic aids.
Related Topics
