Observational Studies Published after the Withdrawal of Rofecoxib
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: NSAIDs - COX-2 Inhibitors – Risks and Benefits
Eleven observational studies on COX-2 inhibitors and adverse cardiovascular outcomes were reported after the withdrawal of rofecoxib and major findings.
OBSERVATIONAL STUDIES PUBLISHED
AFTER THE WITHDRAWAL OF ROFECOXIB
Eleven
observational studies on COX-2 inhibitors and adverse cardiovascular outcomes
were reported after the withdrawal of rofecoxib and major findings are
summarized in Table 47.2. Reports in abstract form or conference proceedings
are not included in this review. The results were heterogeneous as there was
much variation in study design, study populations, comparison groups, and
outcomes of interest. Four reports were based on administrative data of private
or public health insurance data in North America. Shaya and colleagues compared
incidence of APTC events among users of COX-2 inhibitors and non-selective
NSAIDs and found no increased risk among users of rofecoxib or celecoxib when
compared with users of non-selective NSAIDs (Shaya et al., 2005).
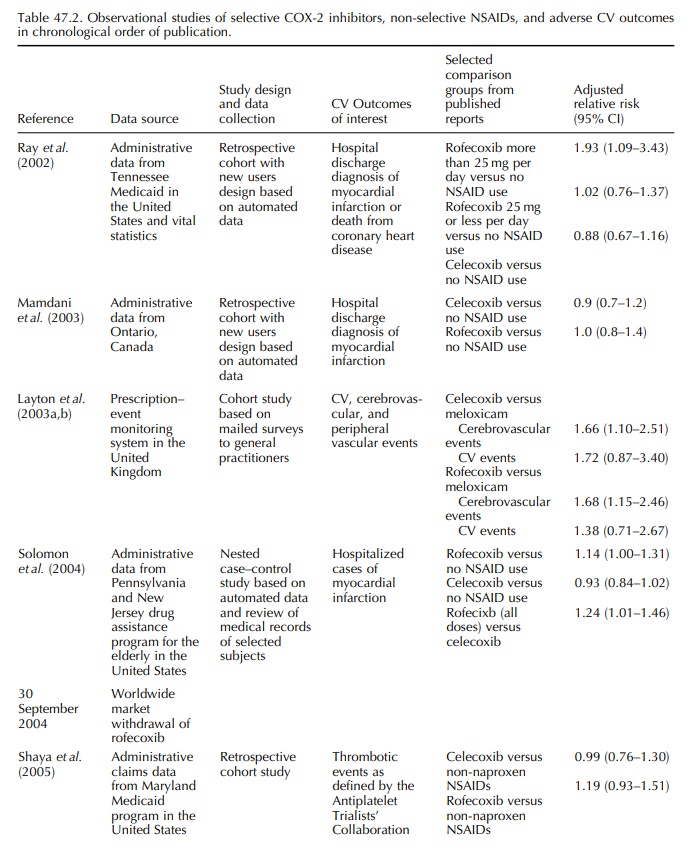
Graham
and colleagues evaluated the risk of myocar-dial infarction and sudden cardiac
death among users of non-selective NSAIDs and COX-2 inhibitors in a nested
case-control study (Graham et al.,
2005). Comparing with those who were unlikely to have used a prescription
non-selective NSAID or COX-2 inhibitor during the last 60 days, the adjusted
odds ratio was 0.84 (95% CI, 0.67–1.04) for celecoxib. Adjusted odds ratio was
higher for high dose rofe-coxib (more than 25 mg per day) than for low dose
rofecoxib (25 mg per day or less). Levesque and colleagues compared current use
of COX-2 inhibitors, non-selective NSAIDs, and no use of either COX-2
inhibitors or non-selective NSAIDs and found that high dose rofecoxib (more
than 25 mg per day) and low dose rofecoxib (25 mg per day or less) were both
associated with increased risk of hospitalized myocar-dial infarction when
compared with no use (Levesque, Brophy and Zhang, 2005). There was no increased
risk among celecoxib users. Velentgas and colleagues reported an increased risk
of acute coronary syndrome among current rofecoxib users compared with
ibupro-fen or diclofenac users (relative risk 1.35; 95% CI, 1.09–1.68) and no
increased risk among current cele-coxib users for the same comparison drugs
(relative risk 1.03; 95% CI, 0.83–1.27) (Velentgas et al., 2006). Huang and colleagues used national health insurance
data from Taiwan and reported no increased risk of myocardial infarction,
angina, stroke, or transient ischemic attack when rofecoxib or celecoxib was
indi-vidually compared with meloxicam among an ethnic Chinese population (Huang
et al., 2006).
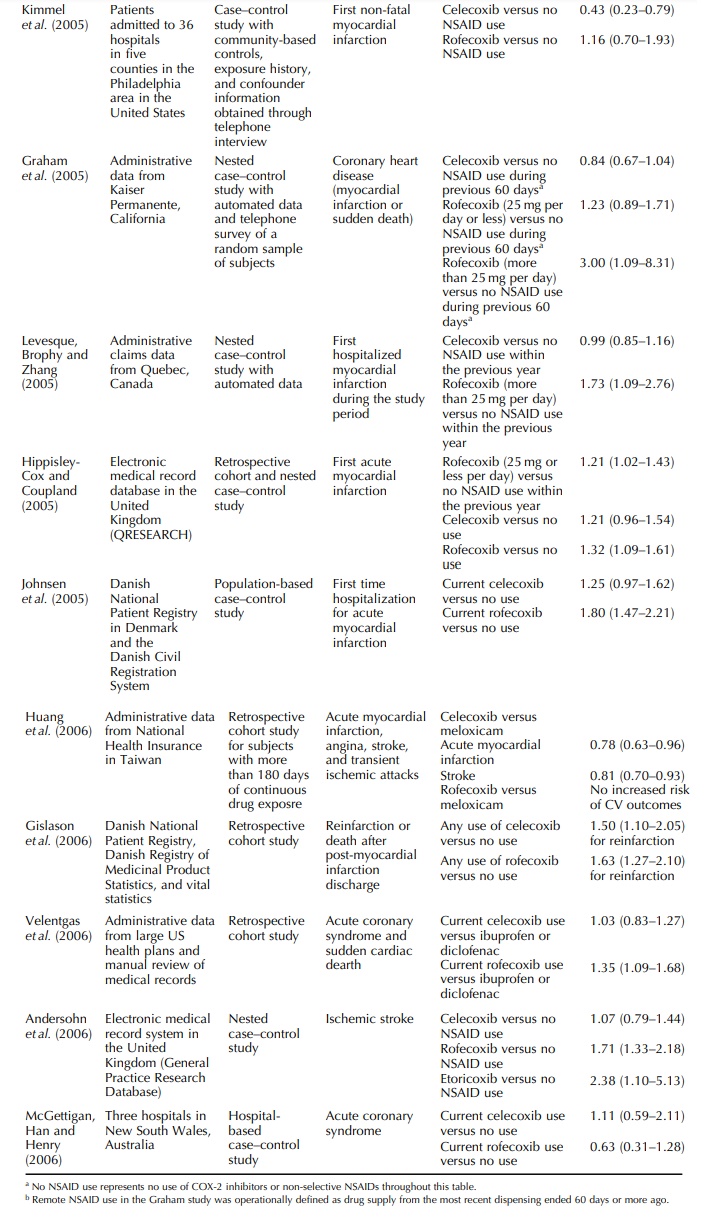
Two
studies were based on electronic medical record systems in the UK.
Hippisley-Cox and Coup-land found that rofecoxib use was associated with
myocardial infarction (adjusted odds ratio 1.32; 95% CI, 1.09–1.61) and celecoxib
use showed similar level of increased risk, but the lower bound of the 95% CI
was 0.97 (Hippisley-Cox and Coupland, 2005). Andersohn studied stroke as an
outcome interest and found increased risk of stroke among rofecoxib and
etoricoxib users but not celecoxib users (Andersohn et al., 2006).
Two
studies were based on population-based registries in Denmark. Johnsen and
colleagues stud-ied the risk of first myocardial infarction and reported
increased risk among rofecoxib users and non-statistically- significant
increased risk among celecoxib users (Johnsen et al., 2005). Gislason and colleagues studied re-infarction and
death after post-myocardial infarction discharge and found increased risk for
both celecoxib and rofecoxib (Gislason et
al., 2006).
The
two other studies were case–control studies with patients identified from
hospitals. Kimmel and colleagues in the US compared the use of COX-2 inhibitors
with no NSAID use and reported adjusted odds ratio of 0.43 (95% CI, 0.23–0.79)
for celecoxib and 1.16 (95% CI, 0.70–1.93) for rofecoxib (Kimmel et al., 2005). McGettigan and colleagues
in Australia studied acute coronary
syndrome and did not find increased risk among celecoxib users or rofecoxib
users (McGettigan, Han and Henry, 2006).
McGettigan
and Henry combined data from 12 observational studies involving COX-2
inhibitors and found that celecoxib was not associated with increased
cardiovascular risk, with a combined rela-tive risk of 1.06 (95% CI, 0.91–1.23)
(McGettigan and Henry, 2006). Both high dose rofecoxib (more than 25 mg per
day) and lower dose rofecoxib (25 mg or less per day) were associated with
increased cardiovascular risk. Combined relative risk for high rofecoxib was
2.19 (95% CI, 1.64–2.91) and it was 1.33 (95% CI, 1.00–1.79) for lower dose
rofecoxib.
Related Topics
