Mechanism of Action Of Insulin
| Home | | Pharmacology |Chapter: Essential pharmacology : Insulin, Oral Hypoglycaemic Drugs and Glucagon
Insulin acts on specific receptors located on the cell membrane of practically every cell, but their density depends on the cell type: liver and fat cells are very rich. The insulin receptor is a hetero-tetrameric glycoprotein consisting of 2 extracellular α and 2 transmembrane β subunits linked together by disulfide bonds.
MECHANISM OF ACTION OF INSULIN
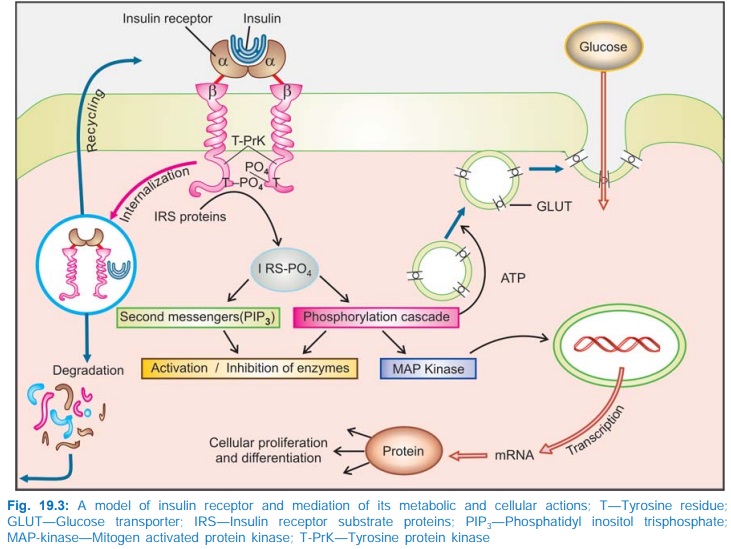
Insulin acts on specific receptors located on the cell membrane of practically
every cell, but their density depends on the cell type: liver and fat cells are
very rich. The insulin receptor is a hetero-tetrameric glycoprotein consisting
of 2 extracellular α and 2 transmembrane β subunits linked together by disulfide bonds.
It is oriented across the cell membrane as a heterodimer (Fig. 19.3). The α subunits carry
insulin binding sites, while the β subunits have tyrosine protein kinase
activity.
Binding of insulin to α subunits induces
aggregation and internalization of the receptor along with the bound insulin
molecules. This activates tyrosine kinase activity of the β subunits → pairs of β subunits
phosphorylate tyrosine residues on each other → expose the catalytic
site to phosphorylate tyrosine residues of Insulin Receptor Substrate proteins
(IRS1, IRS2, etc). In turn, a cascade of phosphorylation and dephosphorylation
reactions is set into motion resulting in stimulation or inhibition of enzymes
involved in the rapid metabolic actions of insulin.
Certain second messengers
like phosphatidyl inositol trisphosphate (PIP3) which are generated
through activation of a specific PI3kinase also mediate the action
of insulin on metabolic enzymes.
Insulin stimulates
glucose transport across cell membrane by ATP dependent translocation of
glucose transporter GLUT4 and GLUT1 to the plasma membrane as well as by
increasing its activity. Over a period of time it also promotes expression of
the genes directing synthesis of GLUT4. Genes for a large number of enzymes and
carriers have been shown to be regulated by insulin primarily through MAP
kinases. Activation of transcription factors also promotes proliferation and
differentiation of specific cells.
The internalized receptor
insulin complex is either degraded intracellularly or returned back to the
surface from where the insulin is released extracellularly. The relative
preponderance of these two processes differs among different tissues: maximum
degradation occurs in liver, least in vascular endothelium.
