Liver-targeted drug delivery
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Organ-specific drug delivery
Liver is the major organ responsible for the metabolism, detoxification, and storage of macromolecules; as well as the production and secretion of bile for digestion.
Liver-targeted
drug delivery
Liver
is the major organ responsible for the metabolism, detoxification, and storage
of macromolecules; as well as the production and secretion of bile for
digestion. It plays an important role in the clearance of pathogens and
antigens entering the body via the GI tract. The need and modalities of
liver-targeted drug therapy is best understood in the context of cellular
components of the liver, the nature of liver diseases, and the cellular
recep-tors on various liver cells that can be utilized for targeted drug
therapy.
Cellular components of the liver
Liver
is designed for the recognition, metabolism, and elimination of for-eign
material, including bacteria, viruses, and noncellular particulates. This role
is served through the anatomical design whereby venous blood is circulated
through the liver, including the parts of the GI tract, via the hepatic portal
vein, through a sinusoidal system.
The
liver consists of four cell types—(1) hepatocyte, (2) endothelial, (3) Kupffer,
and (4) stellate cells. The main parenchymal tissue of the liver is composed of
hepatocytes, which make up 70%–85% of
the liver mass and are involved in various liver activities including the
formation and secretion of bile. Hepatocytes have metabolic, endocrine, and
secretory functions. Liver endothelial cells form the discontinuous lining of
the sinusoids and have fenestrations that are ~100 nm in diameter. This
relatively large pore size plays an important role in determining the sizes of
particles filtering between the blood and the liver parenchymal cells. A space
of Disse sepa-rates hepatocytes from the sinusoids.
The
hepatic sinusoids are lined with the Kupffer
cells, which are the largest group of tissue macrophages in the liver.
Their main function is to phagocytose and destroy foreign material, such as
bacteria or colloids.
Hepatic
stellate cells (HSCs) localize within the space of Disse in close proximity of
both hepatocytes and endothelial cells (Figure 15.4a).
HSCs are present in the perisinusoidal space, constituting about 5%–10% of the
total number of liver cells. These are the mesenchymal cells that are
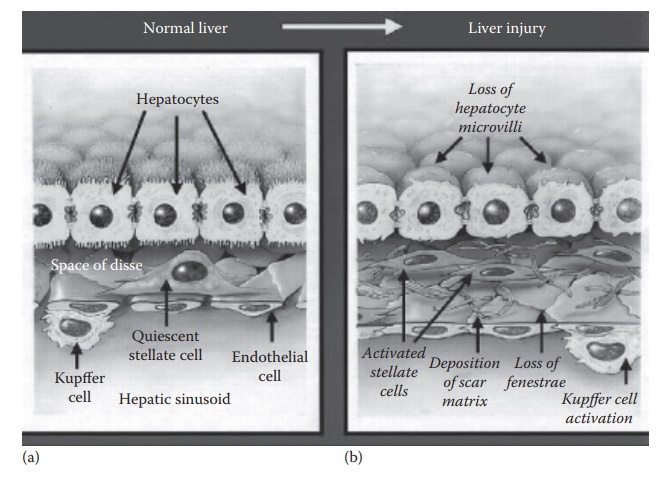
Figure 15.4 Physiology of the (a) normal and (b) diseased liver showing subsinusoidal
events during liver injury. In response to liver injury, stellate cells secrete
excessive extracellular matrix (ECM), which deposits in the subsinusoidal space
of Disse as scar matrix and loss of fenestrae. Liver injury also causes Kupffer
cell activation, which contribute to paracrine activation of stellate cells.
(Reproduced from Friedman, S.L., J. Biol. Chem., 275(4), 2247–2250, 2000. With
permission.)
Stellate cell activation transforms them
into myofibroblasts, cells that are phenotypically between a fibroblast and a
smooth muscle cell. The myofibroblasts produce fibrinogen, a glyco-protein
involved in blood coagulation. Deposition of fibrinogen in the liver can lead
to liver fibrosis.
Common diseases of the liver
The
normal physiology of the liver is affected in the disease state. For example,
in response to liver injury, stellate cells secrete excessive extracel-lular
matrix (ECM), which deposits in the subsinusoidal space of Disse as scar matrix
and loss of fenestrae. Liver injury also causes Kupffer cell activation, which
contribute to paracrine activation of stellate cells (Figure
15.4b). The excessive ECM secretion contributes to the loss of
hepatocyte microvilli and sinusoidal epithelial fenestrae, which leads to loss
of liver function.
Drug
delivery to the liver is indicated in several diseases. For example,
1.
Hepatocellular
carcinoma:
Hepatocellular carcinoma (HCC) is the third
leading cause of cancer-associated deaths worldwide. HCC has been associated
with hepatitis B and C infections, metabolic liver dis-eases, and nonalcoholic
fatty liver diseases.
2.
Cirrhosis: Activation of HSCs
can lead to the deposition of fibrotic tissue.
Continuation of the fibrotic process can lead to end-stage liver disease known
as cirrhosis. Liver cirrhosis is associated with anatomical alteration of the
sinusoidal architecture, reduced liver perfusion, compromised liver function,
and increased risk of HCC. Liver cirrhosis is mainly caused by hepatitis B and
C infections, alco-hol abuse, biliary problems, and fatty liver
(steatohepatitis).
3.
Hepatitis: Hepatitis is a
state of inflammation of the liver that is com-monly caused by viruses, which
are of five main types A, B, C, D, and E. Hepatitis virus types B and C are the
most prevalent, lead to chronic diseases, and are the most common cause of
liver cirrhosis and cancer. Hepatitis virus types A and E are spread by
contaminated food and water. Hepatitis virus types B, C, and D are spread by
parenteral contact with infected body fluids by mechanisms such as injec-tion,
infusion, sexual contact, and mother-to-baby transmission at the time of birth.
Natural mechanisms of hepatic drug uptake
The
natural role of the liver in protecting the body from xenobiotics pro-vides
mechanisms that allow passive drug targeting to the liver. These include the
following:
1. Hepatic
first-pass effect: Orally absorbed drugs are carried through the hepatic portal vein into the liver
before they reach the systemic circulation. The liver metabolizes several drugs
(e.g., diazepam and morphine) to a significant extent, leading to reduced oral
bioavail-ability. This phenomenon can also be utilized for liver targeting
through
a. High hepatic exposure of orally administered compound.
For example, antiviral drugs targeted for the treatment of hepatitis C, such as
ribavirin and telaprevir are administered orally.
b.
Prolonging the circulation time of compounds targeted for the liver provides
passive targeting to the hepatocytes through prolonging the duration of time a
therapeutic is available for hepatocyte uptake. For example, PEGylated
interferons α-2a (PEGASYS®) and α-2b (PegIntron®) have been used
effectively in the treatment of hepatitis B and C in combination with
ribavirin.
2. Enhanced
permeation and retention effect: Liver tissue in diseases such as hepatocellular carcinoma (HCC)
displays the enhanced per-meation and retention (EPR) effect. The EPR effect is
attributed to the imperfect endothelium of neovasculature (newly formed blood
vessels) of growing tumors that result in larger particulate drug carriers
being able to concentrate in the tumor tissue more than the normal tissue with
mature vasculature. This mechanism can be utilized for drug delivery to the
liver by the utilization of particulate drug carriers such as liposomes and
nanoparticles.
Cellular targets for disease therapy
Natural
xenobiotic scavenging role of the liver cells through endocytotic and specific
target/antigen-binding receptors on various cell types affords opportunities
for actively targeted drug therapy for various liver cell types. The receptors
that can be utilized for drug targeting to specific liver cells include the
following:
1. Hepatocytes: They are involved
in liver diseases such as hepatitis A, B,
or C; alcohol-induced or nonalcohol-induced steatohepatitis (NASH); and
genetic diseases such as Wilson’s disease and hemochromatosis. Hepatocytes can
be targeted through the asialoglycoprotein receptors on their cell surface,
which bind galactose and lactose.
a. Asialoglycoprotein receptors on hepatocytes are
attractive as a target receptor for drug delivery because of limited
distribution of these receptors elsewhere in the body, high binding affinity
with the target ligand (e.g., galactose), and rapid ligand internalization.
Galactosylated drug carriers (i.e., DDSs that display galactose res-idues on
their surface) are readily delivered to hepatocytes due to the relatively wide
sinusoidal gap (~100 nm diameter). Drug deliv-ery carriers that are modified
with galactose or lactose have been utilized for drug delivery in HCC.
b. HCC cells also express several growth factor receptors,
such as the epidermal growth factor receptor (EGFR). Antibodies against such
growth factor receptors, such as the anti-EGFR antibody cetuximab have shown
some activity against HCC.
c. Coxsackie- and adenoviral-receptor and integrin receptors
on their cell surface that help internalize adenoviruses. Adenoviral vectors
can be utilized to deliver genes. Hepatocyte selectivity of viral gene delivery
can also be achieved from viral vectors that are derived from the human
immunodeficiency virus (HIV) and the Sendai virus.
d.
Apolipoprotein E is rapidly cleared from the systemic circulation by
hepatocytes. The apolipoprotein E or the high-density lipid (HDL) particles has
been utilized for the delivery of short-interfering RNAs (siRNAs) and microRNAs
(miRNAs).
2. Kupffer cells are highly
phagocytic cells that are a part of the reticu-loendothelial system (RES), also
called the mononuclear phagocyte system (MPS) or the macrophage system. Kupffer
cells can be targeted through a variety of ways. For example,
a. Sugar (mannose and fucose) receptors that serve to
recognize natural foreign particles, such as bacteria and yeast, can be
uti-lized to target proteins and drugs to the RES phagocytic cells. For
example, mannose-modified human serum albumin (HSA) selec-tively accumulates in
Kupffer cells.
b. Kupffer cells phagocytose noncellular particles of 100 nm
or higher diameter. Passive targeting to these endocytotic cells can,
therefore, be achieved using particulate drug delivery carriers.
c. Kupffer cells and endothelial cells express scavenger
receptors, which predominantly bind negatively charged molecules. Proteins and
liposomes with a net negative charge have been utilized for targeting the
scavenger receptors. For example, coupling of the electroneutral dexamethasone
to HSA through lysine residues increases the net negative charge on HSA,
increasing its poten-tial uptake by the scavenger receptors. In addition,
incorporation of succinyl-HSA (HSA conjugated with the polyanionic succinic
acid) into liposomes has been targeted for drug delivery to the sinusoidal
liver endothelial cells.
HSCs
are involved in the fibrotic processes that can lead to liver cirrhosis. In the
presence of chronic liver injury, HSCs get activated and transform into
proliferative myofibroblasts, which are the major source of excessive ECM. The
receptors that are highly upregulated on HSCs include the following:
1. Mannose-6-phosphate (M6P)/insulin-like growth factor II
receptor
2. Collagen type VI receptor
3. Platelet derived growth factor-β (PDGF-β) receptor.
Conjugation
of HSA to M6P or peptides that recognize the collagen type VI or the PDGF-β receptor has been utilized to
target HSCs. These carri-ers have been utilized for the delivery of
antifibrotic small-molecule drugs, proteins, siRNAs, and triplex-forming
oligonucleotides (TFOs) through direct conjugation with the carrier molecules,
complexation/conjugation with carrier molecule-modified HSA, or incorporation
in liposomes that have been modified with the carrier molecule.
Related Topics
