Database Searches and Data Retrieval
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Medical Dictionary for Regulatory Activities (MedDRA)
Here we are concerned particularly with the identification of cases of related medical conditions.
DATABASE SEARCHES AND DATA
RETRIEVAL
Here we are concerned particularly with the identification of cases of related medical conditions. In this respect, MedDRA provides some features that assist in the process, and also some challenges. Database searches and retrieval of data are performed for several purposes, including the review of safety data such as at the end of a clinical trial, evalua-tion of possible safety signals, responding to medical information requests or regulatory authority enquiries about safety and so on. The search strategies and methods used to search for and retrieve the data might be different depending on the intended use of the output.
In
general, it is the Preferred Term that is the focus of searches of safety
databases. However, the categori-sation of these within MedDRA under primary
SOC and then under HLGT and HLT assists in finding rele-vant cases according to
medically relevant groupings. The fixed link between the PT downwards through
the LLT and hence to the case that was originally the subject of the report
provides the mechanism for identifying and retrieving the cases.
The
multiaxial structure of MedDRA helps the user find terms related to the medical
concept being searched for by presenting the terms in more than one SOC
location, should this be appropriate medi-cally. For example, a search of a
database for terms relevant to cardiac failure might reasonably focus on the Cardiac disorders SOC. If a multiaxial
search is performed, this would additionally find PTs for various dyspnoeas
under the HLT Dyspnoeas and HLGT Cardiac disorders signs and symptoms
even though their primary location is in the Respiratorydisorders SOC. Likewise, PTs for Oedema and Peripheral oedema
are found in their secondary loca-tion in the Cardiac disorders SOC as well as in their primary location under General disorders and admin-istration site
conditions SOC.
However,
it is essential to remember that terms in the Investigations SOC (and also those in the Social circumstances and Surgical and medical procedures SOCs) do
not (at present) have secondary locations in other SOCs. It is therefore
necessary to look under those SOCs if relevant terms are not to be missed. It
is also important to keep in mind that multiax-ial locations in MedDRA are an
aid to case finding and data retrieval but they may not be comprehensive
(Brown, 2003).
It
is the very attribute of MedDRA that is most useful for coding – its high
specificity and large size – that presents challenges for database searches and
case retrieval. For example, a table showing adverse events for a product might
be presented as PTs under primary SOC location. For a large database, a
print-out of this table might run to many pages. Selecting the PTs relevant to
a particular medical condition might be quite difficult, if these are only
presented in alphabet-ical order (Brown and Douglas, 2000). In addition, it
would be necessary to look at several SOCs – includ-ing Investigations SOC.
It
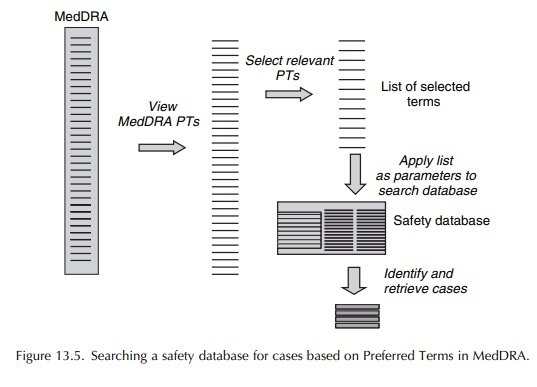
may therefore be useful to show the PTs under the appropriate HLTs and HLGTs,
in order to break down large tables into relevant groupings. An exam-ple is
shown in Table 13.6. However, care still needs to be taken not to miss relevant
terms. As an example, in searching for cases relevant to depression, looking in
the Psychiatric disorders SOC, it
might be tempt-ing to limit a search to PTs found under the HLGT Depressed mood disorders and disturbances and
its subordinate HLTs Depressive disorders
and Mood alterations with depressive symptoms. However, rele-vant terms (and
hence cases) might also be found coded with terms under the HLGT Adjustment disor-ders (incl subtypes),
such as Adjustment disorder with
depressed mood: or some terms under the HLGT Suicidal and self-injurious behaviours NEC. In addi-tion, there
could be PTs relevant to depression under the HLGT Chemical injury, overdose and poisoning in the Injury, poisoning and procedural complica-tions SOC.
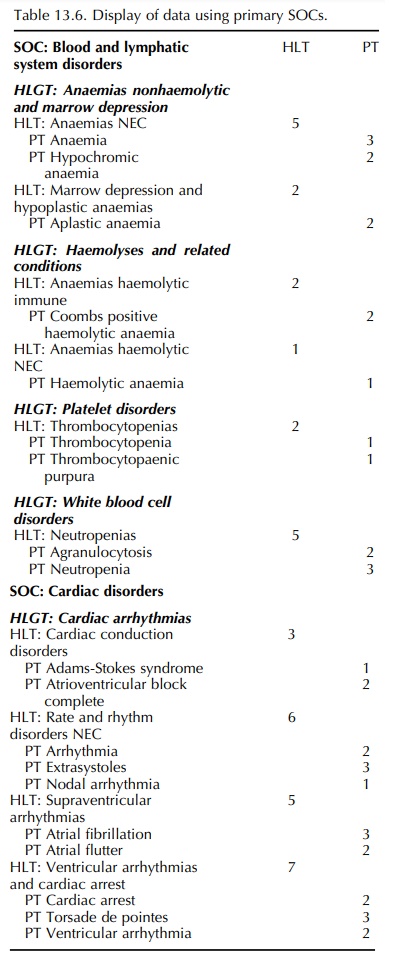
The
type of search referred to above is illustrated in Figure 13.4. It is based on
identifying relevant MedDRA PTs that have been included in a specific database
– for one product, from one source, covering a specified time period (Brown,
2003). As such, the resulting list of PTs cannot be used for searching a
database for another product or for the same prod-uct on a different database,
or for the same database at a different time. In any of these situations,
addi-tional relevant PTs could be present in the database concerned that may
not have been included in the initial search.
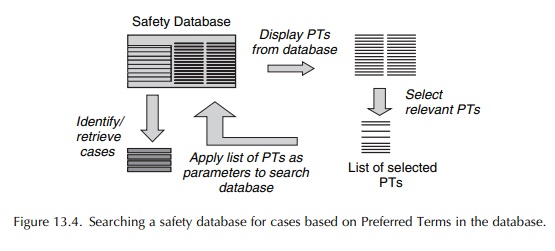
An
alternative approach to searching the database is shown in Figure 13.5. Here,
the search is based on a list of terms derived from the whole of MedDRA, rather
than just derived from the database concerned (Brown, 2003). There is available
a limited number of such searches within MedDRA itself – the Special Search
Categories (SSCs). At the time of writing, there are just 13 of these,
comprising lists of PTs relat-ing to medical conditions and each spanning
several SOCs. Examples are lists of PTs for Haemorrhage, Hypersensitivity
reactions and Cardiac ischaemia. In an initiative that the author was
instrumental in estab-lishing, a CIOMS working group is preparing a series of
such searches – Standardised MedDRA Queries (SMQs) – which will eventually span
the most impor-tant topics for pharmacovigilance. Thus, for example, at the
time of writing, there are SMQs for Rhabdomy-olysis/myopathy; Torsades de
pointes/QT prolonga-tion; Hepatic disorders; Haemolytic disorders; Acute renal
failure; and Severe cutaneous adverse reactions and approximately another 70
are planned. The SMQ searches are made available to MedDRA subscribers only.
Standardised
MedDRA Queries differ from Special Search Categories in that they may include
more than one level of MedDRA, for example a list of PTs and a list of HLTs
(with all their subordinate PTs). They include searches of differing degrees of
speci-ficity and sensitivity – thus, broad searches and asso-ciated narrow
searches comprising subsets of these (SMQs, 2005). An example of an SMQ is
shown in Figure 13.6.

Guidelines
on data retrieval and presentation (presently draft, but endorsed by ICH) have
been produced by a group including representatives from Industry, Regulatory
authorities and the MSSO. The key elements concerning data retrieval are
outlined below .
·
The way that legacy data have been converted to MedDRA might
have an impact on subsequent searches of the database. (This will be discussed
in the next section).
·
Careful documentation of data retrieval methods is essential
for the interpretation of results.
·
Retrieval strategies should be reviewed by a person with a
medical background who is trained in the use of MedDRA.
·
When basing searches on group terms – HLGTs and HLTs – users
should review the terms within these groups to ensure that they are all suited
to the search under consideration.
· Clinically related PTs might be overlooked or not recognised as belonging together as they might exist in different locations within a single SOC. For example, if searching for terms relating to gastric haemorrhage, these might be expected to be present under the HLGT Gastrointestinal haemorrhages NEC, under the HLT Gastric and oesophageal haemorrhages. However, the PTs Haematemesis and Melaena are found under the HLT Non-site specific gastrointestinal haemorrhages, under the same HLGT, whilst the PT Gastric ulcer haem-orrhage is present under the HLT Gastric ulcers and perforation under the HLGT Gastrointestinal ulceration and perforation. Failure to look under this HLGT might lead to underestimation of the number of relevant reports.
·
Users should be aware of primary SOC assignment rules that
will affect the way data are distributed across the terminology. For example,
terms that refer to congenital conditions are located primarily in the Congenital disorders SOC and the
secondary location is the body site. For example, the PT Heart disease congenital has Congenital disorders as the primary and Cardiac disorders as the secondary SOC location. Similar rules
apply for neoplasms and for infections: the location of the disease is assigned
the secondary SOC location.
·
Clinically related PTs in MedDRA might be over-looked or not
recognised as belonging together because they can be distributed among two or
more SOCs. The most important instances are proba-bly those concerning
investigation findings and associated medical conditions. For example, Blood glucose decreased is assigned to the Investigations SOC. Hypoglycaemia
on the other hand is present in the Metabolism
and nutrition disorders SOC.
·
Data may also reside in SOCs that are not antic-ipated
intuitively by the user and, as with the Investigations SOC, multiaxiality may
not apply. For example, in reviewing cases of life-threatening ventricular
arrhythmias, the Cardiac disorders
SOC would be the main candidate for a search for relevant terms. In addition,
terms for sudden death have a primary location in the General disorders SOC, with
a secondary location under Cardiac
disorders SOC. There could also be rele-vant terms for cardiac surgery and
other interven-tions in the Surgical and
medical procedures SOC, and ECG abnormalities in the Investigations SOC. It is important that these last two SOCs and
the Social circumstances SOC should
not be forgotten in searches, in view
of the absence of multiaxial linkages.
·
It is acknowledged that all possible secondary SOC
assignments for a given concept may not be present in MedDRA, but it is possible
to request new link-ages from the MSSO.
The
Guidelines refer to the SSCs and SMQs as options for searching safety
databases. They also high-light the possible effects of changes in version of
MedDRA on search strategies. The version used in the search should be
documented and users should acquaint themselves with the changes that have been
made between versions. The terms used for construct-ing the searches should be
in the same MedDRA version as the data being queried. It is possible that a
search based on an old version of MedDRA might not include all the relevant
terms in a database that is based on a more recent version. Any queries that
are stored for future use should be updated to the appro-priate version of
MedDRA prior to use on new data.
Related Topics
